The aging of the reproductive systems has broad implications on the well-being of the entire organism. Particularly, the onset of female reproductive aging contributes to the development of other age-associated dysfunctions, such as cardiovascular problems, osteoporosis, and muscular and neurological declines. These progressive deteriorations of physiological functions go hand in hand with the changes in bioactive lipid signaling, such as changes in the function of the endogenous cannabinoid system (ECS) and in the levels of circulating steroid hormones.
Interestingly, it appears that the endocannabinoid and steroid systems are closely connected as sex steroid hormones have been found to regulate ECS. Recent research indicates that ECS is active in the ovaries, where it affects egg maturation. Several studies also indicate that the levels of endocannabinoids decrease during aging, while the activity of enzymes that break endocannabinoids down- increases.
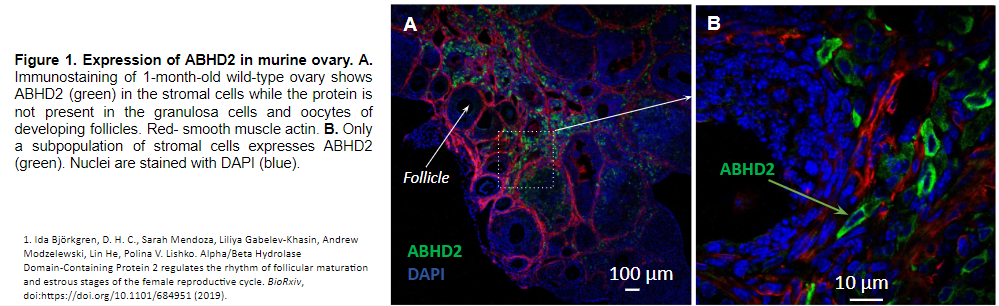
We recently characterized one of these enzymes, termed α/β hydrolase domain- containing protein 2 (ABHD2). Unlike any other enzymes in this family, ABHD2 breaks down endocannabinoids only when it is activated by steroid progesterone. Thus, due to its unusual functional properties as an unconventional progesterone receptor, ABHD2 links together two major signaling systems: endocannabinoids and sex steroids. Interestingly, ABHD2 is highly expressed in the ovarian stroma (1) – the tissue with a well-established role in the ovarian aging.
Here, we propose to further investigate the role of ABHD2 in ovarian physiology and elucidate the signaling pathways by which ABHD2 exerts its action, as well as determine the changes in bioactive lipid profiles that occur with age in the ovaries. The project will utilize transgenic mouse models, genome editing, state-of-the-art electrophysiology, imaging, and mass spectrometry approaches. Based on the exceptional evolutionary conservancy of ABHD2 we can successfully use the mouse as an animal model in our research.


