Progesterone is a steroid hormone that plays an important role in reproductive biology. Spermatozoa encounter Progesterone in the female reproductive tract where it is secreted by the cumulus cells surrounding the egg.
In Lishko et al., 2011, it was shown that in human sperm, Progesterone activates CatSper, the principal Ca2+ channel. Since sperm are transcriptionally and translationally silent, progesterone must achieve CatSper activation through binding to a plasma membrane receptor rather than a nuclear one.
In Miller et al., 2016, we showed that progesterone achieves this non-genomic activation of CatSper by binding to ɑ/ꞵ-hydrolase domain protein 2 (ABHD2), a serine hydrolase that is highly expressed in the cell membranes of sperm flagella. Upon binding to progesterone, ABHD2 cleaves 2-arachidonoylglycerol, an endocannabinoid and a CatSper inhibitor found in the plasma membrane, into glycerol and arachidonic acid. By inducing ABHD2 to cleave 2-arachidonoylglycerol, progesterone thus activates the CatSper channel.
Progesterone is the only steroid present in vivo in the female reproductive tract known to activate the CatSper channel in this manner. (In Mannowetz et al., 2017, we showed that pregnenolone sulfate, not endogenous to the female reproductive tract, also activates CatSper via ABHD2). Other steroids – including testosterone, estradiol, and hydrocortisone – have in fact been shown by our lab to interfere with progesterone’s activation of the CatSper channel in human sperm. This discovery suggests that non-progesterone steroids or the structurally similar triterpenoids could act as contraceptives through interference with progesterone activation of CatSper.

Molecular structure of Progesterone (Image from Wikipedia)
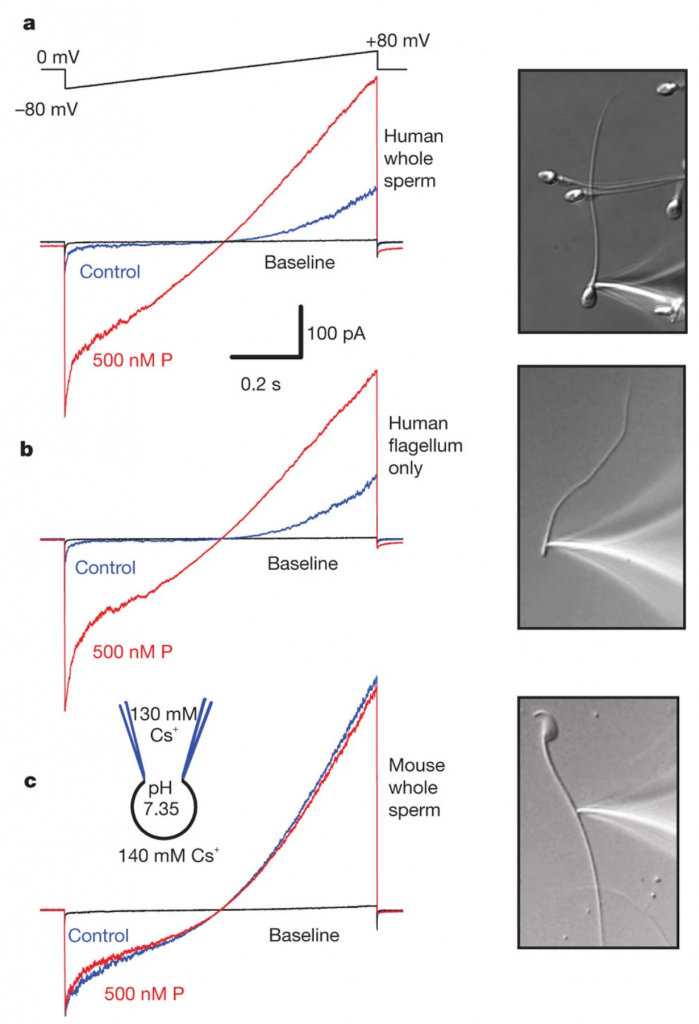
Figure from Lishko et al., 2011

